Writing Balanced Equations Chem 10 Review Worksheet With Answers
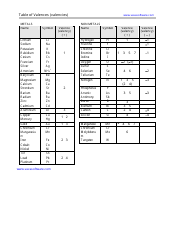
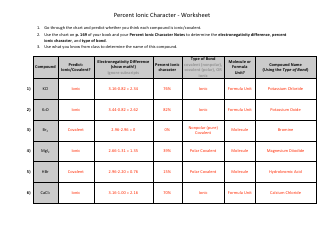
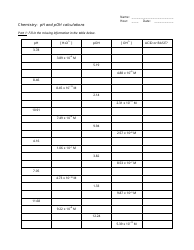
The "Writing Balanced Equations Chem 10 Review Worksheet With Answers" is a study resource used by students in Chemistry 10 to practice and review the skill of writing balanced chemical equations. This worksheet provides a series of chemical reactions for students to analyze and write the corresponding balanced equations. The included answers help students check their work and assess their understanding of the topic.
The Writing Balanced Equations Chem 10 Review Worksheet with Answers is typically filed by the students or their teacher. It is a common practice to keep these types of worksheets as part of the students' educational records.
FAQ
Q: How do you write a balanced chemical equation?
A: To write a balanced chemical equation, you need to ensure that the number of atoms of each element is the same on both sides of the equation. This is achieved by adjusting the coefficients in front of the chemical formulas.
Q: What is the purpose of balancing chemical equations?
A: Balancing chemical equations is important because it ensures that the law of conservation of mass is followed. In other words, it ensures that matter is neither created nor destroyed during a chemical reaction.
Q: Can you provide step-by-step instructions on how to balance a chemical equation?
A: Steps to balance a chemical equation include:
Q: Are there any general rules or strategies to help balance chemical equations?
A: Yes, here are a few strategies:
Q: What are coefficients in a balanced chemical equation?
A: Coefficients are the numbers written in front of the chemical formulas in a balanced equation. They represent the relative amounts of each substance involved in the reaction.
Q: Can you provide an example of a balanced chemical equation?
A: Sure! Here's an example: 2H2 + O2 → 2H2O. In this equation, the coefficients show that two molecules of hydrogen gas react with one molecule of oxygen gas to form two molecules of water.
Q: Why is it important to include state symbols in chemical equations?
A: State symbols (such as (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous solution) provide information about the physical state of the reactants and products. This is important because the state of matter can affect the reaction conditions and the overall outcome.
Q: Are there any exceptions to the law of conservation of mass when balancing chemical equations?
A: No, the law of conservation of mass must always be obeyed when balancing chemical equations. The total mass of the reactants must be equal to the total mass of the products.