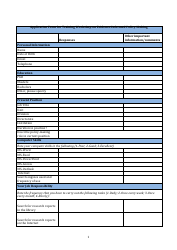
Informed Consent Form for Treatment(S)
The Informed Consent Form for Treatment(s) is used to protect patients' rights by providing them with information about the proposed treatment(s), including potential risks and benefits. It ensures that patients are fully informed and willingly agree to proceed with the recommended treatment(s).
The patient or their legal representative usually files the informed consent form for treatment.
FAQ
Q: What is an informed consent form for treatment?
A: An informed consent form for treatment is a document that explains the benefits, risks, and alternatives of a medical treatment, and is signed by the patient or their representative to indicate that they understand and agree to undergo the treatment.
Q: Why is an informed consent form important?
A: An informed consent form is important because it ensures that patients are fully informed about their treatment options, including the potential risks and benefits, and allows them to make an educated decision about their healthcare.
Q: What should be included in an informed consent form?
A: An informed consent form should include a detailed explanation of the treatment, potential risks and benefits, alternative treatments, possible complications, and the patient's right to revoke consent.
Q: Who needs to sign an informed consent form?
A: The patient or their legally authorized representative needs to sign an informed consent form.
Q: Can a patient refuse to sign an informed consent form?
A: Yes, a patient has the right to refuse to sign an informed consent form. However, it is important for the patient to understand the potential consequences of refusing treatment.
Q: Is an informed consent form legally binding?
A: Yes, an informed consent form is legally binding, and it serves as evidence that the patient provided informed consent for the treatment.
Q: Do all medical treatments require an informed consent form?
A: Not all medical treatments require an informed consent form. In emergency situations or when the patient lacks decision-making capacity, consent may be obtained in other ways.
Q: Can an informed consent form be revoked?
A: Yes, an informed consent form can be revoked at any time by the patient. The revocation should be communicated to the healthcare provider in writing.