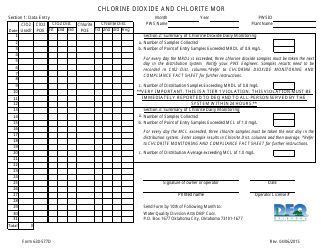
DEQ Form 410-GLD Generally Licensed Device Site Registration - Oklahoma
What Is DEQ Form 410-GLD?
This is a legal form that was released by the Oklahoma Department of Environmental Quality - a government authority operating within Oklahoma. As of today, no separate filing guidelines for the form are provided by the issuing department.
FAQ
Q: What is DEQ Form 410-GLD?
A: DEQ Form 410-GLD is the Generally Licensed Device Site Registration form used in Oklahoma.
Q: What is a Generally Licensed Device?
A: A Generally Licensed Device is a device that meets certain criteria and is authorized for use under a general license.
Q: What is the purpose of the Generally Licensed Device Site Registration?
A: The purpose of the registration is to notify the Oklahoma Department of Environmental Quality (DEQ) about the presence of generally licensed devices at a site.
Q: Who needs to submit DEQ Form 410-GLD?
A: Any person or organization in Oklahoma that possesses or uses a generally licensed device is required to submit the form.
Q: What information is required on DEQ Form 410-GLD?
A: The form requires information about the device, its location, and the responsible party.
Q: Is there a fee for submitting DEQ Form 410-GLD?
A: No, there is no fee associated with submitting the form.
Q: Are there any penalties for not submitting DEQ Form 410-GLD?
A: Failure to submit the form can result in enforcement action and penalties by the DEQ.
Q: Is DEQ Form 410-GLD only required in Oklahoma?
A: Yes, DEQ Form 410-GLD is specific to Oklahoma and its requirements may differ in other states.
Form Details:
- Released on January 1, 2017;
- The latest edition provided by the Oklahoma Department of Environmental Quality;
- Easy to use and ready to print;
- Quick to customize;
- Compatible with most PDF-viewing applications;
- Fill out the form in our online filing application.
Download a fillable version of DEQ Form 410-GLD by clicking the link below or browse more documents and templates provided by the Oklahoma Department of Environmental Quality.