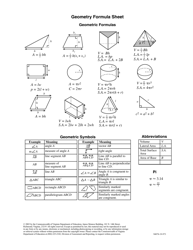
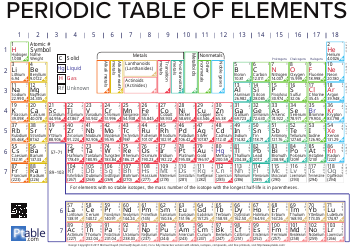
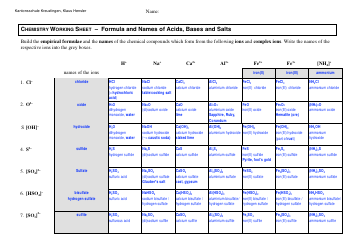
Advanced Chemistry Formulas Cheat Sheet
The Advanced Chemistry Formulas Cheat Sheet is a document that provides a quick reference guide for commonly used chemistry formulas and equations. It is often used by students and professionals in the field of chemistry to help them remember and apply these formulas in their work.
FAQ
Q: What is a cheat sheet for advanced chemistry formulas?
A: A cheat sheet is a quick reference guide for advanced chemistry formulas.
Q: Why would someone use an advanced chemistry formulas cheat sheet?
A: Someone might use a cheat sheet to quickly access and review complex chemistry formulas.
Q: What is the purpose of an advanced chemistry formulas cheat sheet?
A: The purpose of a cheat sheet is to help students or professionals quickly recall and apply complex chemistry formulas.
Q: What types of formulas are typically included in an advanced chemistry formulas cheat sheet?
A: An advanced chemistry formulas cheat sheet may include equations for organic chemistry, physical chemistry, inorganic chemistry, and more.